The bathroom mirror is still slightly fogged from your morning shower, and the faint, sharp scent of citrus fills the space. Outside, the morning frost grips the glass, holding the Canadian winter at bay while you stand barefoot on the cold tile. This is your quiet five minutes before the day demands your attention. You reach for the amber glass bottle on the shelf, unscrewing the pipette with a practiced twist.
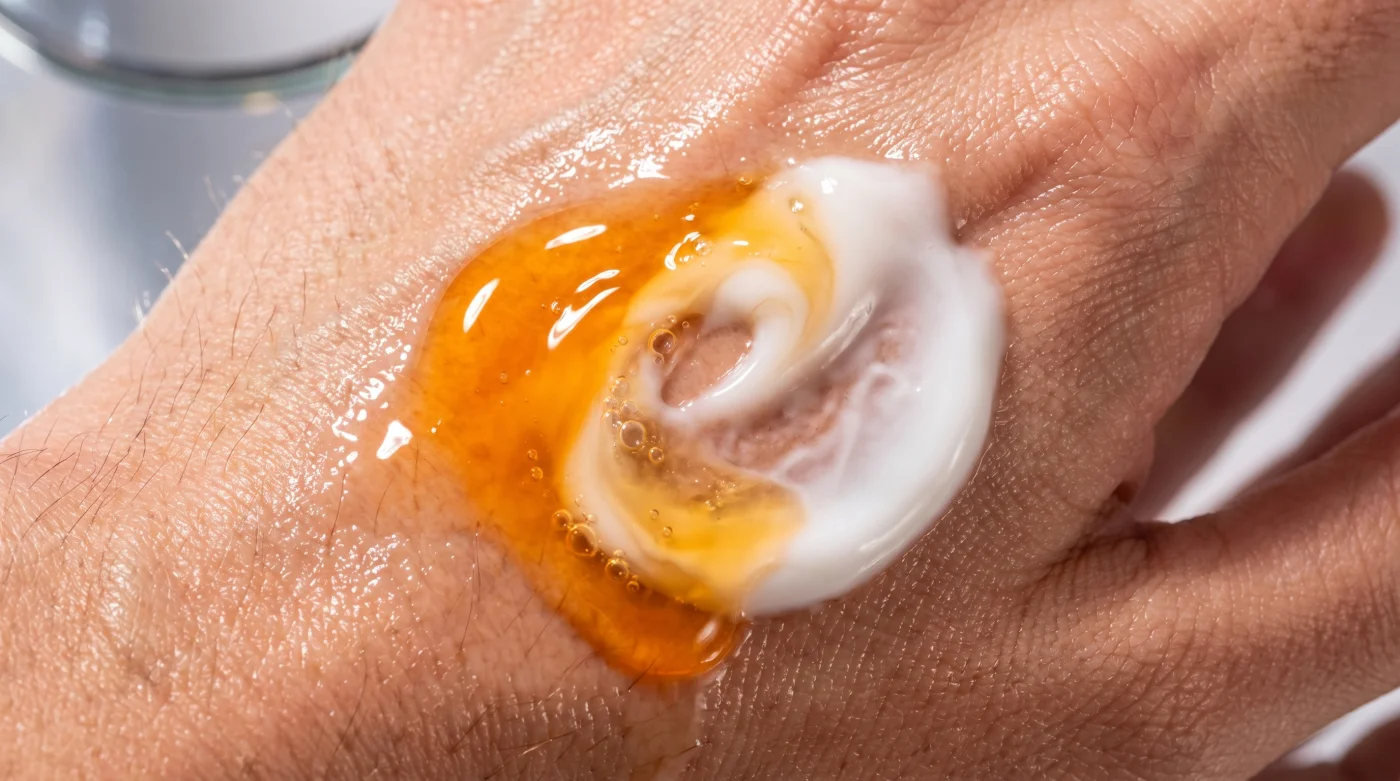
You press the dropper, carefully dispensing liquid gold into your palm. It is an expensive investment, a tiny vial of hope promising bright, firm skin. You press it gently into your cheeks and forehead, feeling the slight, familiar tackiness. Immediately, to save time and lock in the moisture, you grab your favourite daily face cream, slathering a generous dollop right over top to finish the routine.
As you rub the white cream into your skin, the textures blend beautifully. It feels like you are doing everything right, checking off the boxes of a responsible morning habit. You wipe your hands on a towel, confident that the active ingredients are sinking deeply into your pores, preparing to defend your face against the harsh winds and dry indoor heating.
But underneath that comforting layer of hydration, a silent chemical collision occurs. Without realising it, your rushing hands have just created a hostile environment on the surface of your face. By immediately applying a standard lotion over a highly unstable acid, you have completely wiped out the very ingredient you paid so much money to use.
The Physics of a Fragile Acid
L-ascorbic acid, the most potent and common form of Vitamin C, is notoriously delicate. Think of it less like a sturdy root vegetable that can withstand hours of boiling, and more like a fragile soufflé that collapses at a sudden change in temperature. It requires a highly specific, acidic environment—usually a pH level between 3.0 and 3.5—to remain stable enough to penetrate the outer lipid barrier of your skin.
When you immediately layer a standard daily cream over it, you drastically alter the pH of your skin’s surface. Most over-the-counter moisturizers are formulated to be pH-balanced to the skin’s natural resting state, hovering comfortably around 5.5 to 7.0. Mixing these two products is like pouring cold milk directly into a pan of hot, sputtering butter. The reaction instantly stops.
Your skin does not suddenly absorb both products independently. Instead, the higher pH of the cream neutralises the low pH of the serum right there on your cheeks. The acid loses its electrical charge, rendering it entirely inactive before it can pass through the stratum corneum. The glow you were expecting is washed away by simple, unavoidable chemistry.
The Formulator in the Cold Room
Dr. Clara Hughes, a 42-year-old botanical chemist based out of a clinical lab in Toronto, sees this mistake daily. Working in a sterile, temperature-controlled room overlooking the icy grey expanse of Lake Ontario, she spends her days testing the degradation rates of antioxidants. “We spend months calibrating the exact acidity needed for L-ascorbic acid to survive,” she notes, tapping a glass beaker. “But then people take this perfectly balanced formula home and mix it with a basic lotion in under ten seconds. They are practically turning a fifty-dollar active compound into expensive water. We formulate in a vacuum, but people apply their products in a hurricane.”
Calibrating Your Specific Canvas
Not every morning looks the same, and neither does every face. The trick is not to abandon your hydration, but to understand how to stagger the application based on what your skin genuinely requires. It is about creating space between the layers.
- The 60-second morning jawline technique that replaces expensive Botox fillers
- The nightly micellar water mistake aging Canadian women a decade faster
- Dermatologists reveal why over-the-counter retinol cream accelerates aging after 45
- Women over 50 are applying hyaluronic acid backward and destroying their skin barrier
- Major scandal reveals hidden PFAS chemicals in Canada’s top lash products
- Health Canada confirms severe new restrictions on this everyday makeup ingredient
- Why luxury skincare brands are quietly removing this filter from their formulas
- The professional application trick that prevents midday makeup melting
- The pantry staple that completely strips product buildup from thinning hair
- The zero-dollar facial technique that mimics professional jawline fillers
For the Purist
If you are someone who thrives on exactness, your goal is to respect the absorption window. Apply your L-ascorbic acid to bone-dry skin immediately after cleansing. Do not apply it to damp skin, as water itself has a neutral pH of 7.0 and can dilute the acid. Wait an entire ten minutes before reaching for your daily cream. Let the acid sink in, do its work, and normalise. Only then should you seal the barrier.
For the Sensitive Soul
Those fighting winter redness face a different challenge. A pH of 3.0 can feel like a stinging bite on cheeks already ravaged by sub-zero winds. If waiting ten minutes leaves your face feeling tight and inflamed, swap the pure L-ascorbic acid for a derivative like sodium ascorbyl phosphate. Derivatives are chemically modified to remain stable at a higher, more comfortable pH of 6.0. They take longer to show results, but they allow you to layer your soothing creams immediately without killing the active ingredient.
For the Morning Rusher
If a ten-minute wait time feels impossible while packing lunches and searching for car keys, you need to rethink the chemistry entirely. Instead of a watery serum, use an anhydrous (water-free) Vitamin C suspension. Because these formulas contain no water, they do not have a pH level. You can apply them, wait a mere thirty seconds, and immediately apply a thicker, oil-based cream on top without triggering a neutralising reaction.
The Tactical Waiting Game
Shifting your routine requires a small, mindful adjustment to the way you manage those early hours. It is not about adding more products to your cabinet; it is about respecting the tools you already own.
Adopt a staggered morning rhythm. Brush your teeth while waiting for the serum to absorb. Make the bed. Pack your bag. Let the product sit undisturbed. When you finally return to the mirror for the final layer of hydration, you will know the acid has already done its job.
- The Temperature Check: Store your pure L-ascorbic acid in a cool, dark place. If it turns dark orange or brown, it has oxidized and is entirely useless.
- The Dry Canvas: Always pat your face completely dry with a clean towel before the acid touches your skin.
- The 10-Minute Rule: Set a mental timer. Give the low-pH formula time to penetrate the lipid barrier before introducing a high-pH cream.
- The Gentle Pat: When you finally apply your daily cream, gently press it into the skin rather than aggressively rubbing it around.
Intentional Rituals Over Empty Layers
There is a profound satisfaction in understanding the subtle mechanics of the things we use every day. When you stop blindly layering products and start considering how they interact, your morning routine shifts from a frantic chore into a deliberate, quiet practice.
You are no longer just putting things on your face; you are orchestrating a reaction. By giving your acid the acidic environment it demands, you honour the investment you made in yourself. You allow the ingredients to work as they were intended, protecting you from the environment, brightening the shadows, and offering a small, daily victory before you even step out the door.
“True skincare is less about the sheer volume of products you stack on your face, and entirely about the respect you show to the chemistry between them.”
| Key Point | Detail | Added Value for the Reader |
|---|---|---|
| The pH Clash | Vitamin C requires pH 3.0-3.5; moisturizers sit at 5.5-7.0. | Stops you from wasting money by neutralizing expensive actives. |
| The Wait Time | A 10-minute pause between acid and cream application. | Ensures deep lipid penetration before the barrier is sealed. |
| The Dry Canvas | Applying acid to completely dry, rather than damp, skin. | Prevents tap water (pH 7.0) from diluting the delicate acid. |
Does waiting 10 minutes actually make a visible difference?
Absolutely. It prevents the rapid pH shift that destroys the acid, allowing the L-ascorbic molecule to fully penetrate the skin barrier.Can I just mix the serum into my cream in my palm?
No. This instantly raises the pH of the acid, neutralizing the active compound before it even makes contact with your face.What if my face gets too dry while waiting?
If the tightness is unbearable, you may have a compromised barrier. Switch to a stable derivative that does not require a harsh low-pH environment.Does keeping my serum in the fridge help?
Yes. Cold temperatures drastically slow down the oxidation process, extending the lifespan of the fragile molecules inside the bottle.Can I skip the moisturizer entirely?
It depends on your local climate and skin type, but in cold, dry Canadian winters, sealing the skin with a lipid layer is highly recommended to prevent water loss.